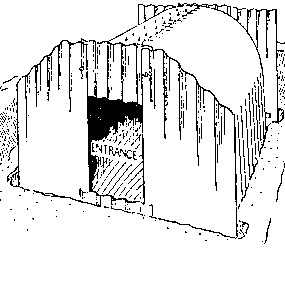
Strong by design

Anderson shelters were very strong – especially against a compressive force such as from a nearby bomb – because of their corrugation. It is relatively easy to bend a flat sheet of metal, but corrugated metal can only easily be bent along the lines of corrugation, and not across them. The roofs of the shelters therefore had to be bent into semi-circular shape in factories and, once bent into shape, they resisted any further inward bending. So the whole structure resisted any further distortion.
Also, the front and rear elements of the shelter were not firmly attached to the two sides, so they could to some extent move independently. This flexibility helped to increase the survival prospects of anyone inside the shelter if a bomb dropped nearby.
Anderson shelters were made of steel, which mainly consists of Iron, and so normally rusts quite quickly in the open air. The steel was therefore coated in Zinc which provided very effective protection against rusting. The process of coating steel with Zinc is called galvanisation and the product was called galvanised steel.
Zinc protects against rusting because it reacts more readily with Oxygen (in air) to form Zinc Oxide. This then reacts with water vapour in the air to form Zinc Hydroxide. Then, finally this compound reacts with Carbon Dioxide in the air to form Zinc Carbonate. And the great thing about Zinc Carbonate is that it is impermeable and insoluble, and sticks very firmly to its parent Zinc and to steel. It therefore forms a very strong barrier which resists any further attack on either the steel or the Zinc coating.
Further protection was provided by painting the galvanised steel and/or burying it under plenty of earth. As a result, some Anderson shelters are in pretty good condition over 70 years later and can be visited.

See also:
- Building your Anderson shelter – the government’s instruction manual etc.
- Restoring and rebuilding – how to go about restoring an Anderson shelter
- Makers marks – who built your Anderson shelter?